Publications
As corresponding author
(15)
Optimization of α-fluoro, β-heteroaryl acrylamide warheads for KRAS G12C active state inhibition
Matthew L. Condakes, Rita L. Civiello, Brian L. Venables, Derek B. Danahy, Richard R. Moore, Srividya B. Balachander, Lisa Chourb, Daniel P. Downes, Dieter M. Drexler, Liudmila Dzhekieva, Miriam El-Samin, Sirish Kaushik Lakkaraju, Christopher G. Levins, Matthew J. Meyer, Katherine Mosure, Michael F. Parker, Jie Qi, Jack L. Sloane, Matthew Soars, Justin Stedman, Nicolas Szapiel, Rebecca L. Thompson, Yong-Jin Wu, Zhuo Zhang, Xiaoliang Zhuo, Michelle L. Stewart, and Joanne J. Bronson
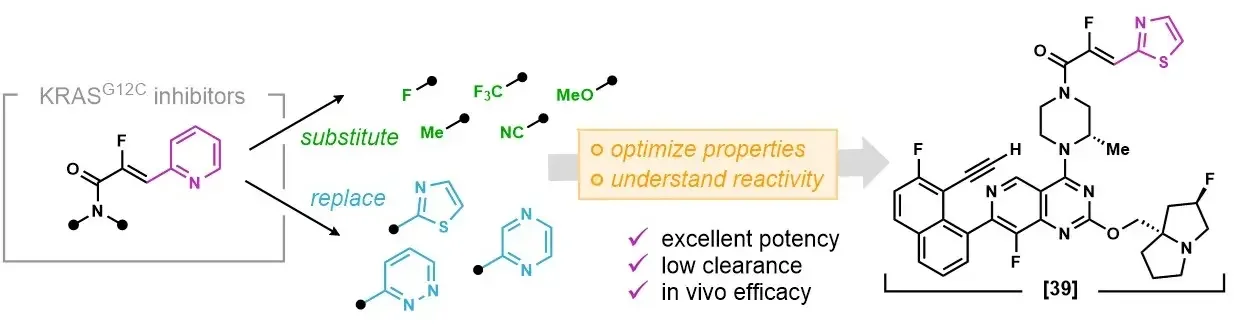
J. Med. Chem. Submitted
(14)
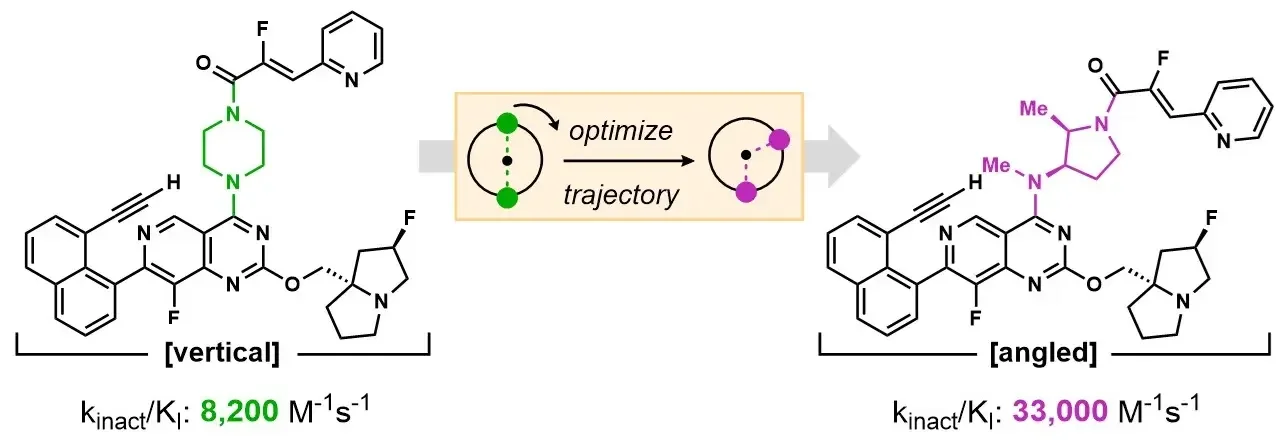
Optimization of covalent warhead trajectory for KRAS G12C active state inhibition
Matthew L. Condakes, Rita L. Civiello, Sirish Kaushik Lakkaraju, Jack L. Sloane, Lisa Chourb, Daniel P. Downes, Dieter M. Drexler, Liudmila Dzhekieva, Miriam El-Samin, Christopher G. Levins, Matthew J. Meyer, Katherine Mosure, Michael F. Parker, Jie Qi, Max Ruzanov, Steven Sheriff, Justin Stedman, Nicolas Szapiel, Rebecca L. Thompson, Zhuo Zhang, Xiaoliang Zhuo, Michelle L. Stewart, and Joanne J. Bronson
J. Med. Chem. Submitted
(13)
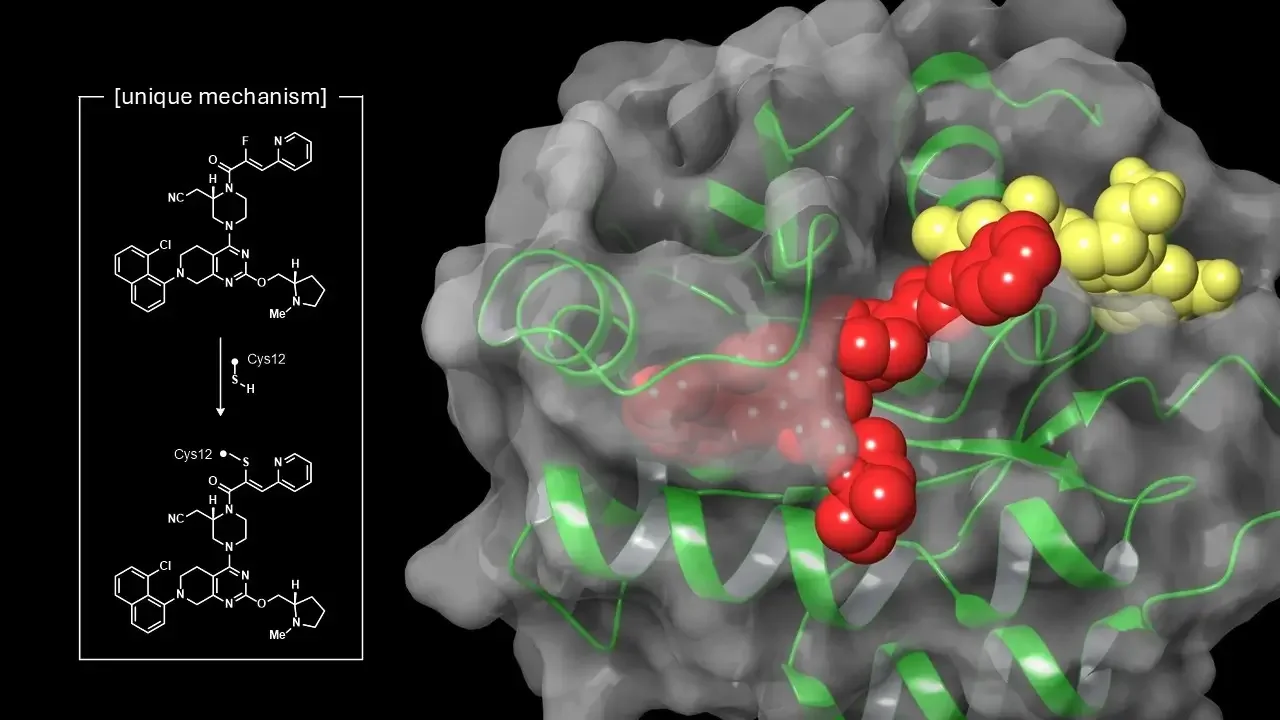
Covalent inhibitor design confers activity against both GDP- and GTP-bound forms of KRAS G12C
Matthew L. Condakes, Zhuo Zhang, Derek B. Danahy, Richard R. Moore, Sirish Kaushik Lakkaraju, Xiaoliang Zhuo, Yuka Amako, Robert M. Borzilleri, Srividya B. Balachander, Lisa Chourb, Rita L. Civiello, Ashok R. Dongre, Daniel P. Downes, Dieter M. Drexler, Brianne M. Dudiak, Liudmila Dzhekieva, Miriam El-Samin, Brian E. Fink, Kosea Frederick, Cherrie Huang, Javed Khan, Emma Lees, Christopher G. Levins, Courtney McCarthy, Gabriel A. Mintier, Katherine Mosure, Michael F. Parker, Ryan Powles, Jie Qi, Max Ruzanov, Sanya Sharma, Steven Sheriff, Ashish K. Singh, Justin Stedman, Nicolas Szapiel, Rebecca L. Thompson, Wayne Vaccaro, Tai Wang, Tianfu Yang, Dan You, Matthew J. Meyer, Joanne J. Bronson, and Michelle L. Stewart
Nat. Comms. Accepted
(12)
New Chemical Entities Entering Phase III Trials in 2022
Matthew L. Condakes, Jennifer Jiang, David W. Lin, Rhiannon Thomas-Tran, Juan del Pozo, and Christiana N. Teijaro
Chapter 23 in 2023 Medicinal Chemistry Reviews, 577-592.

(11)
New Chemical Entities Entering Phase III Trials in 2021
Matthew L. Condakes, Anne E. Hurtley, David W. Lin, Rhiannon Thomas-Tran, and Juan del Pozo

(10)
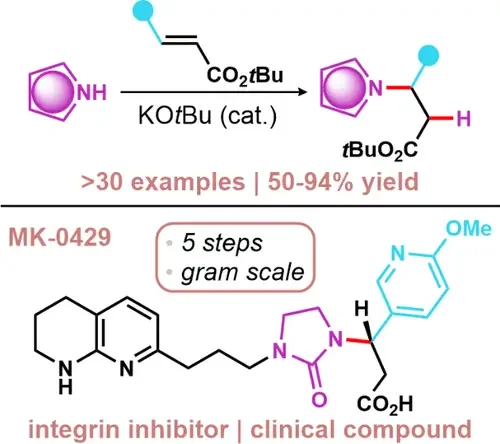
Thermodynamic Understanding of an Aza-Michael Reaction Enables Five-Step Synthesis of the Potent Integrin Inhibitor, MK-0429
Anya Gupta, and Matthew L. Condakes
J. Org. Chem. 2022, 86, 17523-17527. Highlighted in Synfacts
(9)
New Chemical Entities Entering Phase III Trials in 2020
Matthew L. Condakes, Alec H. Christian, Anna E. Hurtley, David W. Lin, Rhiannon Thomas-Tran, and Karl Haelsig
Chapter 20 in 2021 Medicinal Chemistry Reviews, 663-681.

(8)
New Chemical Entities Entering Phase III Trials in 2019
Matthew L. Condakes, Anna E. Hurtley, David W. Lin, Gregory T. Notte, Vickie Tsui, Nathan E. Wright, and Alec H. Christian
Chapter 26 in 2020 Medicinal Chemistry Reviews, 607-623.

Supervised work
(7)
Development of a Terpene Feedstock-based Oxidative Synthetic Approach to the Illicium Sesquiterpenes
Kevin Hung, Matthew L. Condakes, Luiz F. T. Novaes, Stephen J. Harwood, Takahiro Morikawa, Zhi Yang, and Thomas J. Maimone
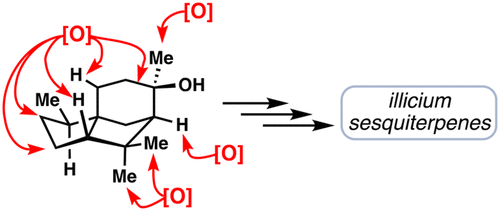
(6)
A Copper-catalyzed Double Coupling Enables 3-Step Entry into the Quassinoid Core Architecture
Matthew L. Condakes, Rachel Z. Rosen, Stephen J. Harwood, and Thomas J. Maimone
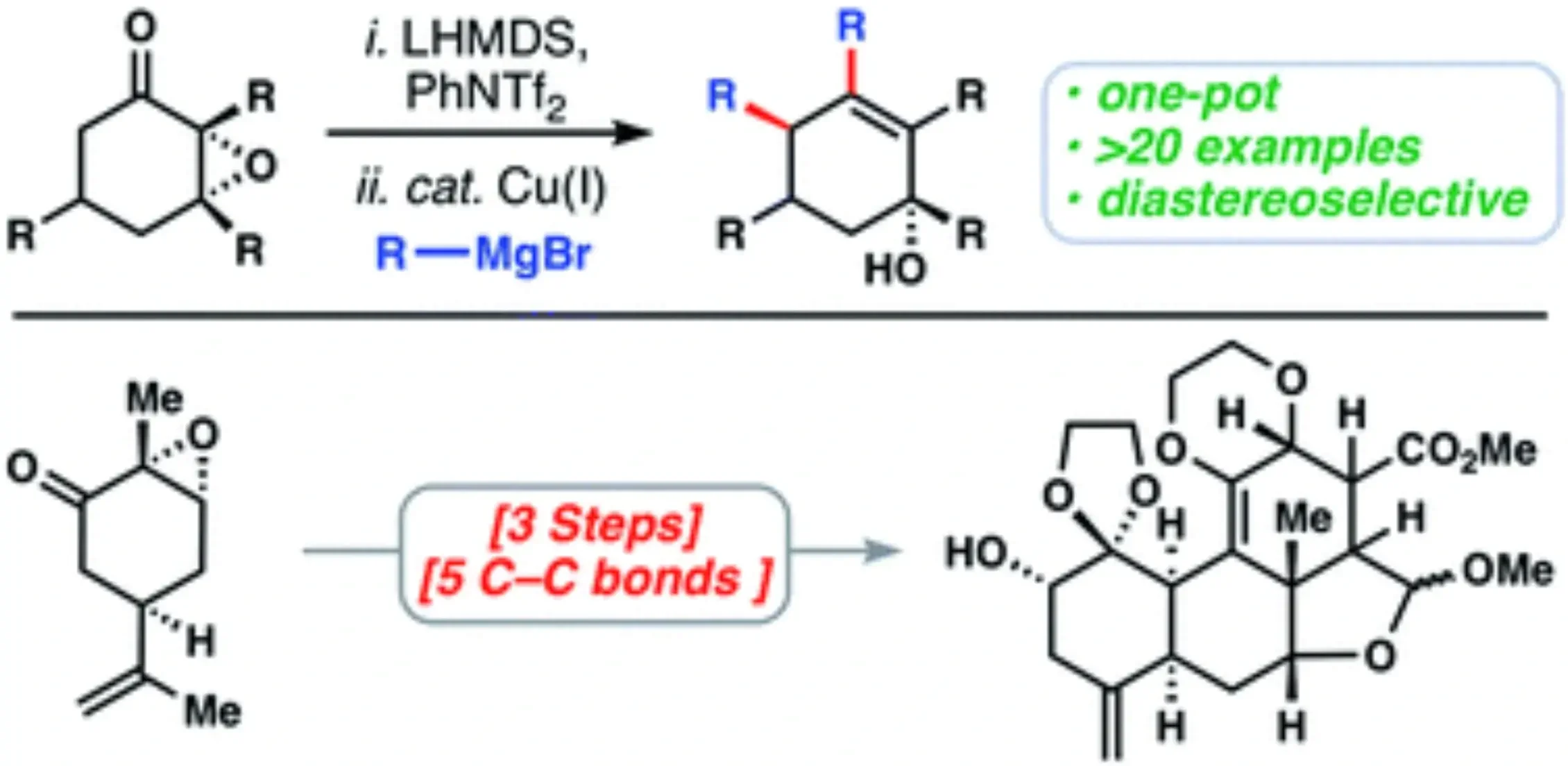
(5)
Contemporary Synthetic Strategies Toward Seco-Prezizaane Sesquiterpenes from Illicium Species
Matthew L. Condakes, Luiz F. T. Novaes, and Thomas J. Maimone
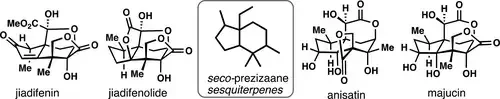
(4)
Total Syntheses of (–)-Majucin and (–)-Jiadifenoxolane A, Complex Majucin-Type Illicium Sesquiterpenes
Matthew L. Condakes, Kevin Hung, Stephen J. Harwood, and Thomas J. Maimone
J. Am. Chem. Soc. 2017, 139, 17783-17786. ACS Editors’ Choice; JACS Spotlight; ACS Select
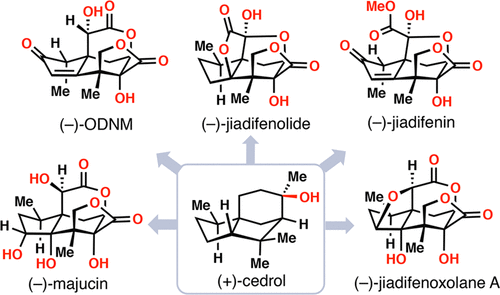
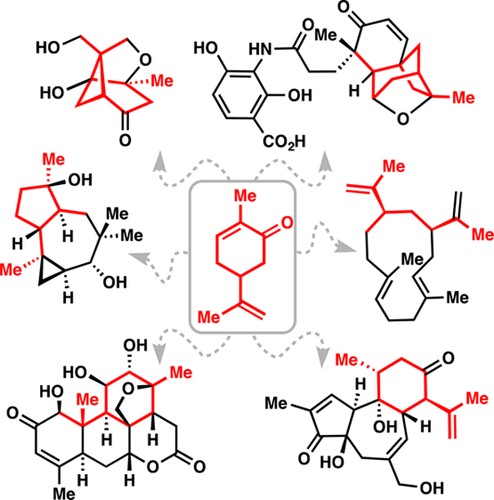
(3)
Navigating the Chiral Pool in the Total Synthesis of Complex Terpene Natural Products
Zachary G. Brill, Matthew L. Condakes, Chi P. Ting, and Thomas J. Maimone
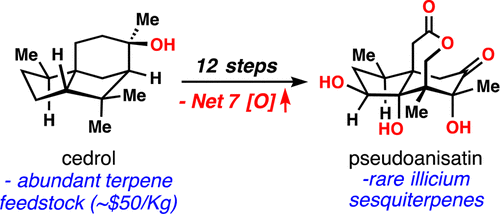
(2)
Oxidative Entry into the Illicium Sesquiterpenes: Enantiospecific Synthesis of (+)-Pseudoanisatin
Kevin Hung, Matthew L. Condakes, Takahiro Morikawa, and Thomas J. Maimone
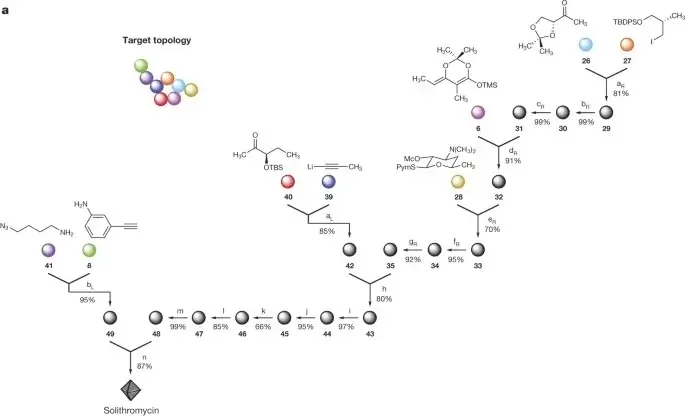
(1)
A platform for the discovery of new macrolide antibiotics
Ian B. Seiple, Ziyang Zhang, Pavol Jakubec, Audrey Langlois-Mercier, Peter M. Wright, Daniel T. Hog, Kazuo Yabu, Senkara Rao Allu, Takehiro Fukuzaki, Peter N. Carlsen, Yoshiaki Kitamura, Xiang Zhou, Matthew L. Condakes, Filip T. Szcypinski, William D. Green, and Andrew G. Myers
Published Patent Applications
(5) Chu, H.; Reisberg, S. H.; Zajdlik, S. H.; Yang, K. S.; Carelli, J. D.; Condakes, M. L.; Harwood, S.; Thompson, P. A. Cysteine covalent modifiers of akt1 and uses thereof. WO2025217336A1, Oct. 16, 2025.
(4) Condakes, M. L.; Civiello, R. L.; Bronson, J. J.; Fink, B. E.; Parker, M. F. KRAS G12C Inhibitors. WO2025122619A1, June 12, 2025.
(3) Condakes, M. L.; Civiello, R. L.; Bronson, J. J.; Parker, M. F. KRAS Inhibitors. WO2024036270A1, February 15, 2024.
Highlighted in ACS Med. Chem. Lett. 2024, 15, 1201.
(2) Condakes, M. L.; Civiello, R. L.; Bronson, J. J. Tetrahydropyrido 3,4-d pyrimidine derivatives as KRAS inhibitors. WO2023240189A1, December 14, 2023.
(1) Condakes, M. L.; Civiello, R. L.; Bronson, J. J.; Parker, M. F. Pyrido[4,3-d] pyrimidine derivatives as KRAS inhibitors. WO2023240188A1, December 14, 2023.